|
If the energies of the two photons are in the ratio of 3 : 2, their wavelengths will be in the ratio of.Electronic configuration of calcium atom can be written as.Uncertainty in position of an electron (mass of an electron is =9.1×10−28g)….4d, 5p, 5f and 6p orbitals are arranged in the order of decreasing energy.According to the Bohr Theory, which of the following transitions in the hydrogen atom will give rise to the least energetic photon?.The energy of second Bohr orbit of the hydrogen atom is −328kJmol−1, hence the energy of fourth Bohr orbit would be. 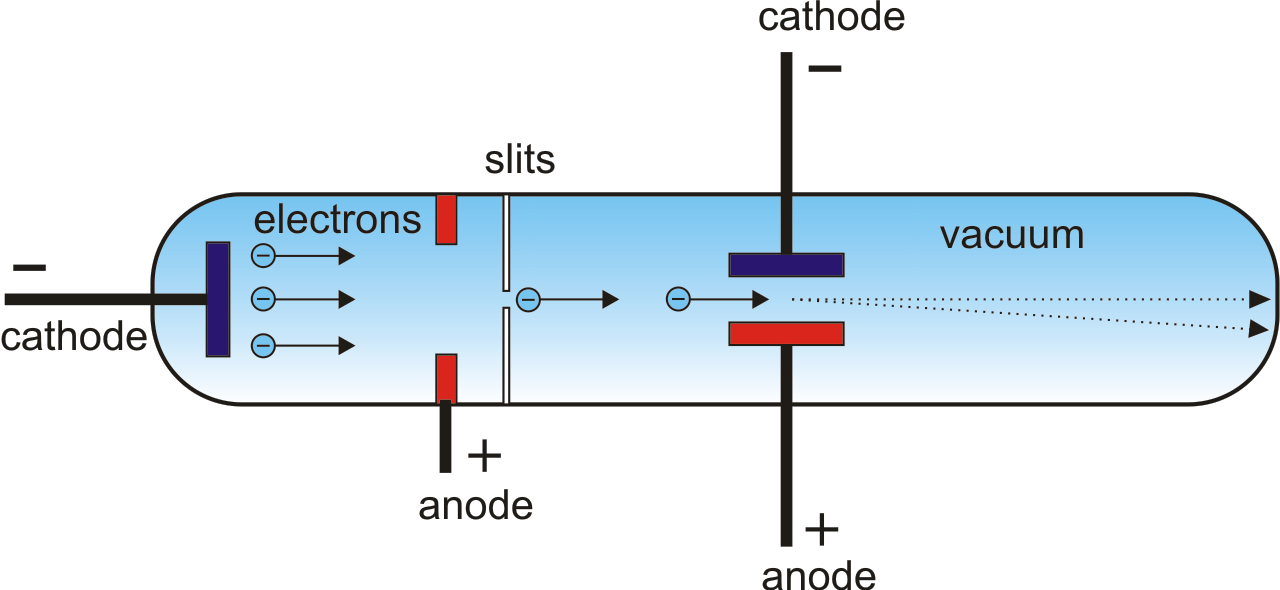
Calculate the energy in joule corresponding to light of wavelength 45 nm.The orbitals are called degenerate when:…. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
